Portable kit makes urine tests more cost-effective
The new test can be carried out at home by using a smartphone and it promises to improve disease prevention while decreasing the cost of urinalysis
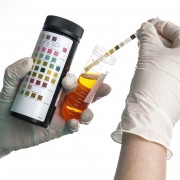
The Israeli company that claims to be behind the world’s first smartphone camera used for clinical-grade diagnostics has just received FDA approval for a kit that enables urine tests to be performed at home on a mobile device.
It is the first time the FDA has ever granted Class II approval for smartphone urine testing, Healthy.io said of its new Dip.io urine test.
With millions of people, including pregnant women, kidney disease sufferers and diabetes patients getting their urine tested every day, the kit’s manufacturers believe billions of dollars could be saved annually on inefficient healthcare spending by enabling more home testing. Indeed, urine testing is the world’s second-most commonly conducted diagnostic test.
“Conducting the same test in the comfort of one's home using a smartphone can improve patient outcomes enormously while potentially saving healthcare expenditures through better access and prevention,” Healthy.io said in a statement.
The kit works by testing 10 parameters, including a range of infections, chronic illnesses and pregnancy-related complications, and is built around the established urinalysis dipstick. The user conducts the test as normal, and then uses their smartphone’s camera to scan the dipper through an app that sends the results to their doctor immediately. Its makers claim that it is as accurate as standard lab-based urinalysis.
Founder and chief executive Yonatan Adiri believes that consumers are in the midst of “the biggest economic value transition in history” with the development of pocket diagnostic systems and the evolution of new healthcare technology.
"One trillion dollars annually of inefficient healthcare spending in America alone is now starting to be captured by technology companies that specialise in transparent value creation for less resources,” he said, adding that his company pursues an “era of the medical selfie that transforms embedded smartphone cameras into clinical-grade medical scanners.”
The approval of the device opens the door for improved screening for kidney disease, which affects over 10 percent of the global population, said Prof Joe Coresh, Professor of Epidemiology at Johns Hopkins and chair of Healthy.io's medical advisory board.
"It's exciting to see the FDA applying its rigour and enabling the use of the smartphone for better patient care."
Related Articles
Will smartphones help diagnose Zika?
The ubiquitous devices might facilitate the diagnosis of the mosquito-borne disease in areas without basic medical infrastructure
Read moreVR could be a boon for anxiety disorder patients
The technology might help patients ease their anxiety by providing exposure to computer-generated frightening environments
Read moreThe battle against ageing
Is a breakthrough that will give us more healthy years on the horizon?
Read moreLatest Articles
Medical Care
Can a Stroke Lead to Dementia? What You Need to Know
Can Stroke Cause Dementia? Learn about the relationship between these two in our insightful article. Discover prevention and management strategies.
Read moreMedical Care
Clearing the Fog: Dispelling Common Diabetes Myths in Singapore
Uncover the truths and myths about diabetes with insights from Harmony Thyroid, Endocrinology and Diabetes Centre. Learn about prevention, diagnosis, and management strategies for diabetes in Singapore. Get expert guidance from Senior Consultant Endocrinologist Dr. Vikram Sonawane to navigate your diabetes journey effectively.
Read moreMedical Care
Achieving Swift Recovery: Enhanced Recovery (ERAS) Direct Anterior Approach Total Hip Replacement
Consider total hip replacement with Alps Orthopaedic Centre's ERAS Direct Anterior Approach for faster recovery and reduced hospital stays. Learn about Dr. Jerry Chen's expertise in Singapore.
Read more